
Oxilithe used in rebreathers and escape apparatus
Oxilithe technology
The origin of the Oxilithe technology began with the scientific work of the French chemist Professor Georges Jaubert, who at the beginning of the 20th century discovered that sodium peroxide in solid form offered an ideal solution for enabling breathing in enclosed spaces. He succeeded in processing this substance into tablets or granules which he gave the commercial name Oxilithe, the unique property being that upon contact with water vapor and carbon dioxide from the exhaled air, the substance entered into a chemical reaction that simultaneously generated fresh oxygen and bound the harmful carbon dioxide gas.
When Robert Henry Davis, then director of the British company Siebe Gorman, caught wind of this revolutionary French invention, he immediately saw the potential for rescue work and military applications where heavy oxygen cylinders were a hindrance. Around 1904, Siebe Gorman acquired the exclusive rights to integrate Jaubert’s technology into their own designs, leading to the development of the first compact Oxilithe rebreathers. These devices were used by the British Navy and in mining as escape and rescue apparatuses, where the user only had to breathe through a mouthpiece into an Oxilithe-filled chamber to provide themselves with an autonomous air supply for a limited time. The close cooperation between Jaubert’s scientific insights and Siebe Gorman’s technical expertise thus laid the foundation for modern chemical breathing apparatuses as we would later see them in aviation and space travel.

The use of Oxilithe in Siebe Gorman’s early rebreathers brought significant safety risks, related both to the aggressive chemical properties of sodium peroxide and to the physiological limitations of breathing pure oxygen under pressure.

Chemical Hazards and “Caustic Cocktails”
The biggest concern with using sodium peroxide was the substance’s extreme reactivity. When Oxilithe came into contact with liquid water—for example, due to a leak in the breathing circuit or excessive condensation—a violent exothermic reaction occurred. This could lead to:
- Chemical burns: The reaction produces sodium hydroxide (caustic soda), a strong alkaline substance that can seriously damage the airways, mouth, and lungs if inhaled or contacted. This phenomenon is known in the diving world as a “caustic cocktail“.
- Explosion hazard: Sodium peroxide is a powerful oxidizer that can spontaneously ignite flammable materials and even explode if stored incorrectly or if heat builds up.
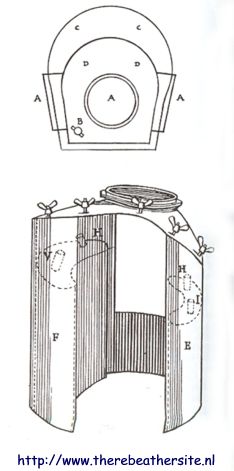
Application in the Hall-Rees-Davis escape set:
The apparatus was developed around 1907-1908 by Captain S.S. Hall, fleet doctor Oswald Rees, and Robert Henry Davis, after which the design was patented on August 18, 1908. The set worked based on a canister with Oxilithe that generated oxygen and absorbed carbon dioxide during breathing. In terms of design, it consisted of a bell-shaped helmet connected to a waterproof tunic with an internal Oxilithe cartridge.

Although the British Royal Navy adopted it as standard escape equipment around 1910 and trained crews in its use, the system was often considered too bulky for the cramped submarines. Moreover, there is no evidence that the device was ever successfully used in a real emergency. Ultimately, the set was replaced in 1927 by the more compact Davis Submerged Escape Apparatus, but the device did gain notoriety as diving equipment in the silent film version of 20,000 Leagues Under the Sea from 1916.


Explanation of how Oxilithe works and its dangers:


Mechanism (How it works):
- Oxygen Generation: The peroxide ions (
) in
are unstable in the presence of moisture and undergo a redox reaction, releasing pure oxygen gas (
).
- CO2 Scrubbing: The compound acts as a “scrubber.” By converting gaseous
into solid sodium carbonate (
), it effectively removes the exhaled carbon dioxide from the air.
- Self-Regulating: Water vapor (from human breath) acts as a catalyst for the initial release of oxygen, making it a highly efficient passive air revitalization system.
*** Why is Sodium Peroxide is dangerous to be used in breathing apparatus: ***
1. Thermodynamic Instability
The peroxide bond () in sodium peroxide is relatively weak and contains oxygen in an unstable oxidation state of -1. In the presence of water, it rapidly seeks a more stable state: the -2 state (in ) and the 0 state (in gas). The transition to these more stable chemical bonds releases a massive amount of energy.
2. High Enthalpy of Reaction
The reaction releases approximately 140 to 150 kJ/mol of heat. The chemical equation is:
This heat is generated so quickly that it can cause the surrounding water to boil or ignite nearby flammable materials.
3. Rapid Formation of Strong Base
The reaction produces sodium hydroxide (), which is a very strong base. The dissolution and hydration of the newly formed
in water is itself an extremely exothermic process, adding even more thermal energy to the system.
4. Gas Expansion
The instantaneous production of oxygen gas () causes rapid expansion. This turbulence increases the surface area contact between the remaining
and the water, leading to a “runaway” effect where the reaction accelerates almost instantly.
Safety Warning: Because of this intense heat and the production of pure oxygen (which supports combustion), sodium peroxide is a fire hazard when it comes into contact with organic materials or moisture.

Therebreathersite was founded by Jan Willem Bech in 1999. After a diving career of many years, he decided to start technical diving in 1999. He immediately noticed that at that time there was almost no website that contained the history of closed breathing systems. The start for the website led to a huge collection that offered about 1,300 pages of information until 2019. In 2019, a fresh start was made with the website now freely available online for everyone. Therebreathersite is a source of information for divers, researchers, technicians and students. I hope you enjoy browsing the content!

Leave a Reply